Patient-Centric Strategy
My Role — UX Strategist & Lead Designer, Mobile Health Initiative · Medidata Solutions · 3 years · Deliverables: mobile health strategy, patient engagement platform, behavioral design framework, user research program, design system
Industry Recognition — Clinical Partnership of the Year · Scrip Award Nomination
The Problem
Clinical trial participation places enormous demands on patients — managing complex medication schedules, completing surveys, attending visits, and staying engaged across months or years — yet the tools built to support them had always been designed for institutions, not people. The result was persistent and measurable: poor adherence, high dropout, and a fundamental disconnect between what trials needed from patients and what patients needed to keep going.
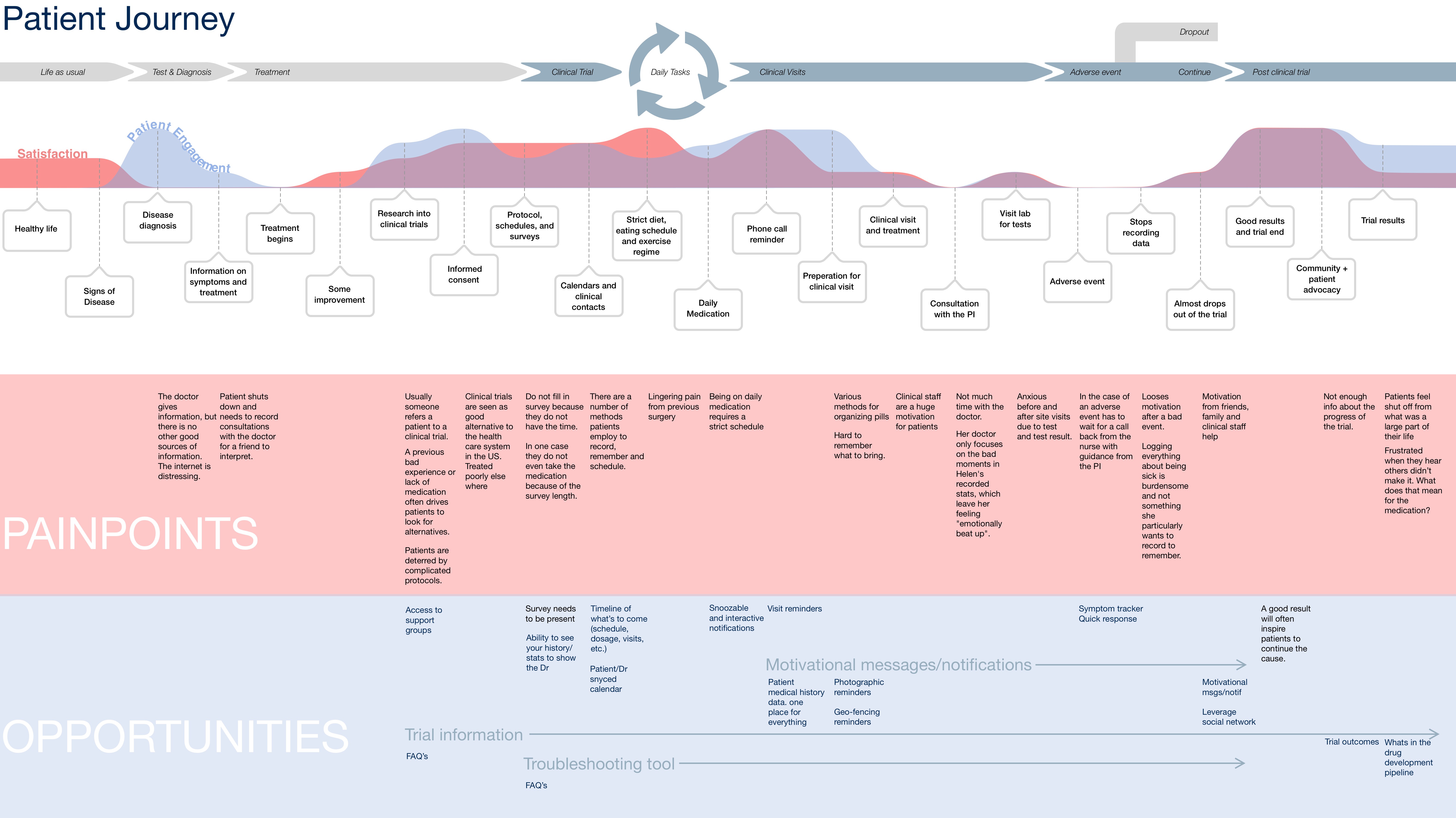
The Research Approach
I designed a research programme that went beyond the industry convention of five-user testing, drawing on evidence that small samples miss significant proportions of real user problems. Research ran across three groups simultaneously: patients managing ongoing medication regimes, site personnel including nurses, physicians, and CRAs, and sponsors and medical directors overseeing large multi-site trials.

From the research I produced affinity maps, user journey maps, detailed archetypes, and a comprehensive app map with feature recommendations. Across all groups, a consistent pattern emerged: patients who sustained engagement had found a personal reason for being in the trial — a North Star — that the existing product did nothing to surface or reinforce.

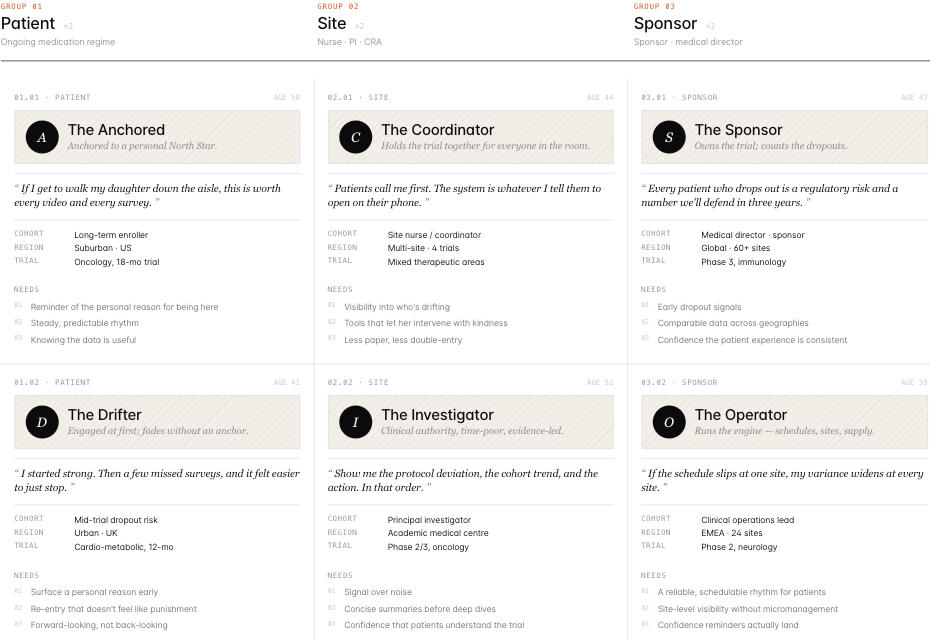
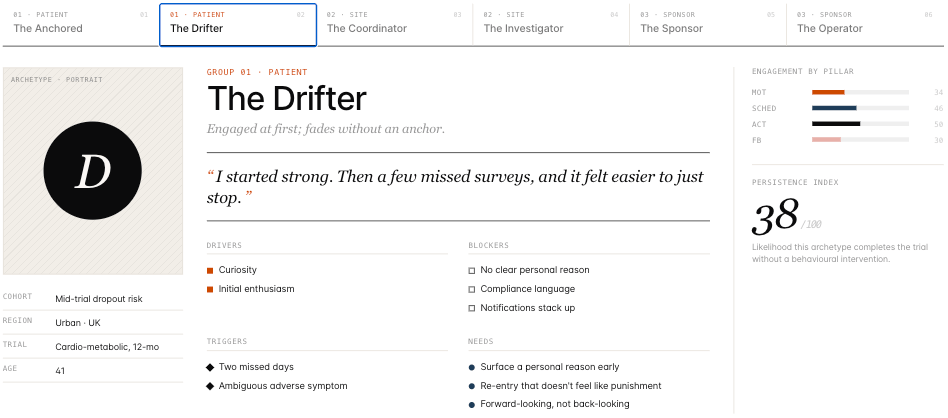
The Design Framework
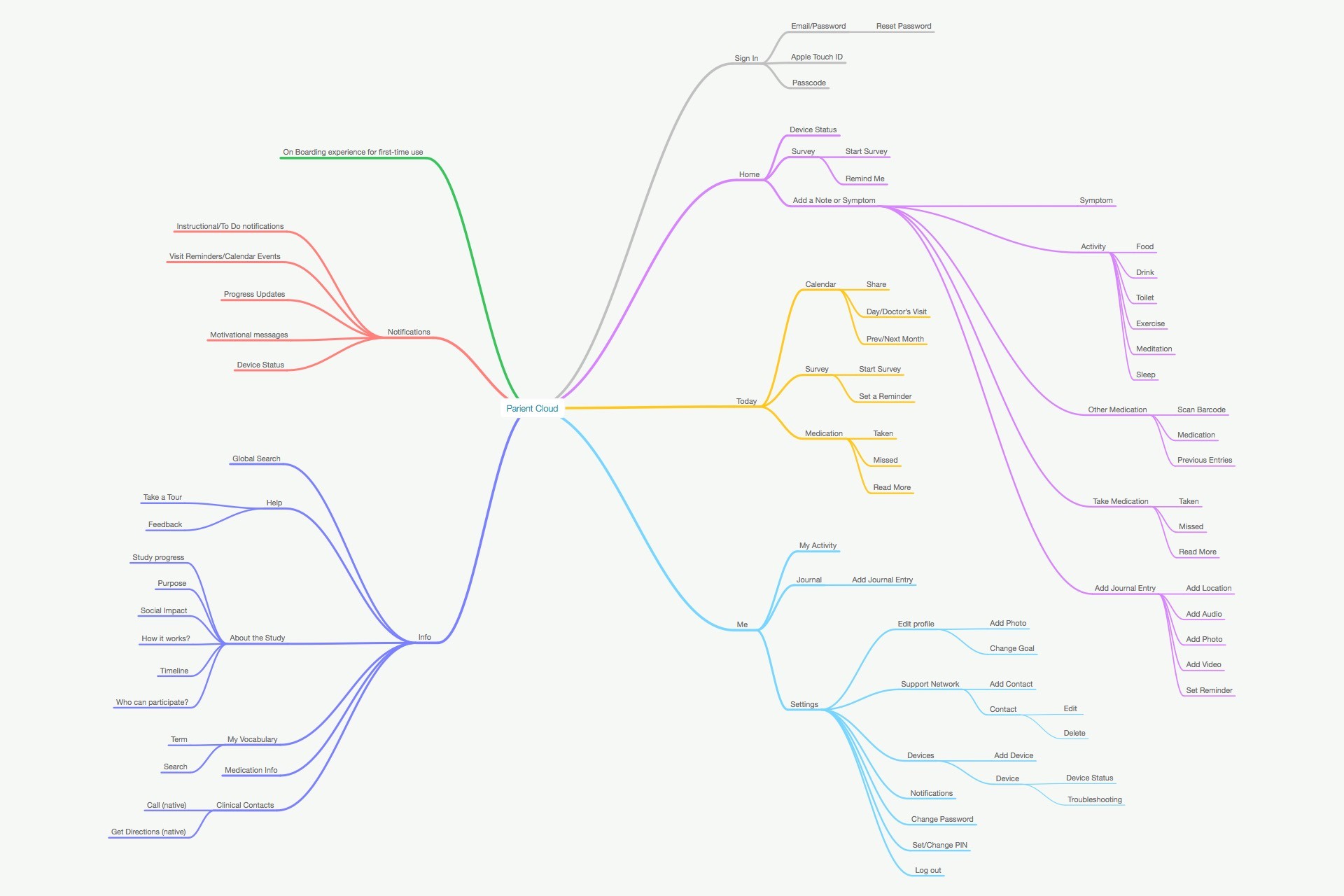
The research produced a behavioural design framework built around four interconnected pillars: Motivation, Scheduling, Activities, and Feedback.
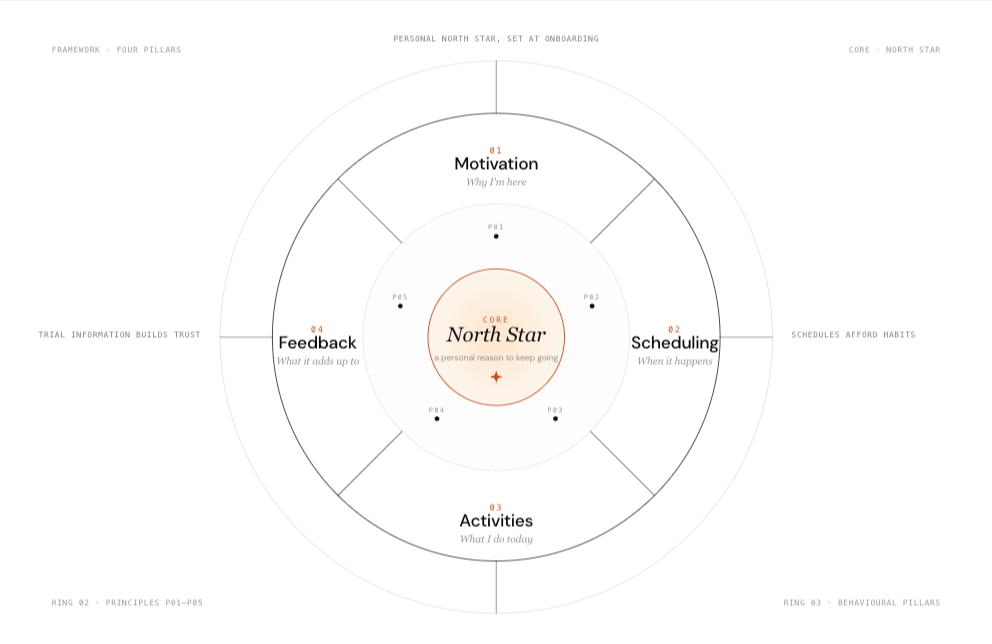
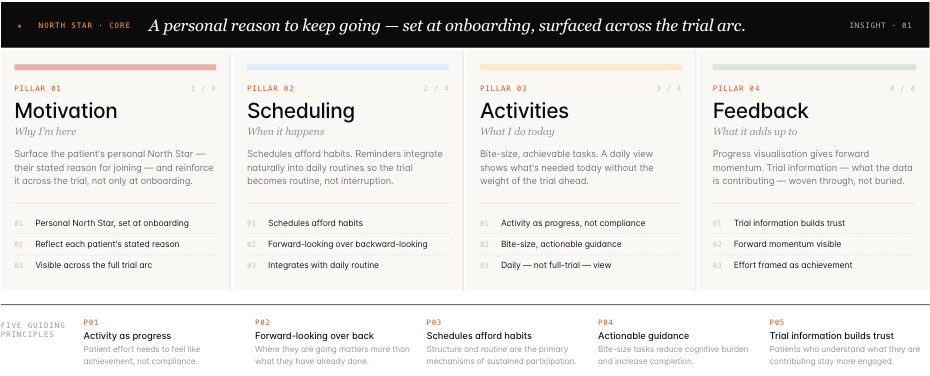
The most important insight was about motivation specifically. Patients who sustained engagement weren't driven by abstract altruism — they had set a personal North Star at the outset. The design needed to surface and reinforce that goal throughout the experience, not just at onboarding. Five additional principles emerged from the research to guide every subsequent design decision:
— Activity as progress: patient effort needed to feel like achievement, not compliance — Forward-looking over backward-looking: where they were going mattered more than what they'd already done — Schedules afford habits: structure and routine were the primary mechanisms for sustained participation — Actionable guidance: bite-size tasks reduced cognitive burden and increased completion — Trial information builds trust: patients who understood what they were contributing stayed more engaged
The Design Response
These principles shaped a mobile experience designed to feel like a supportive companion rather than a monitoring tool. The app centred on a daily task view that gave patients a clear, achievable picture of what was needed from them that day — without overwhelming them with the full weight of the trial ahead.
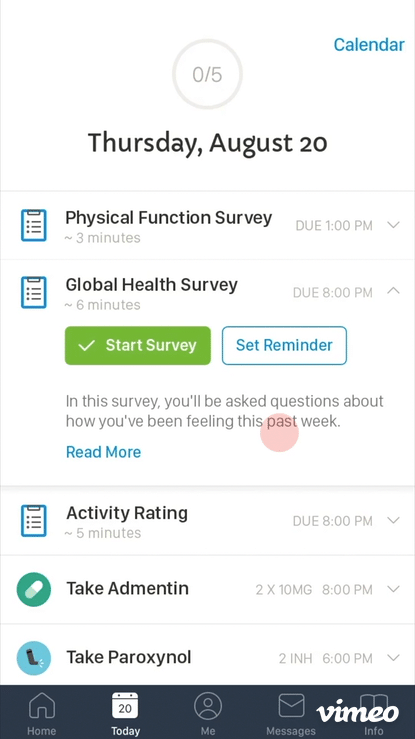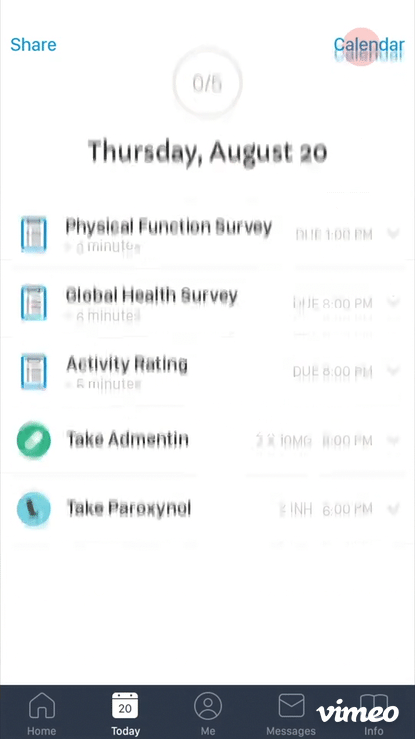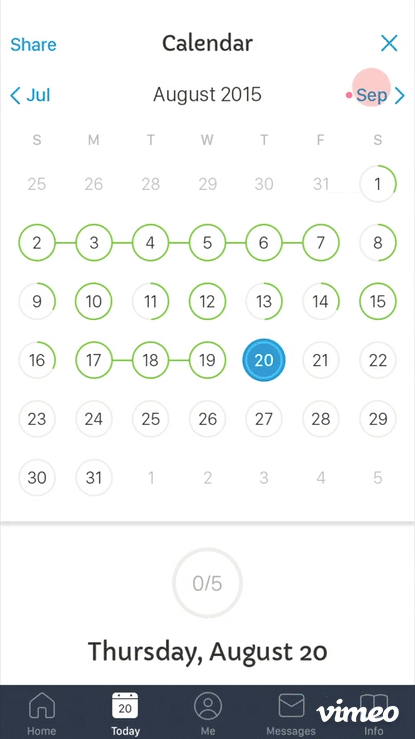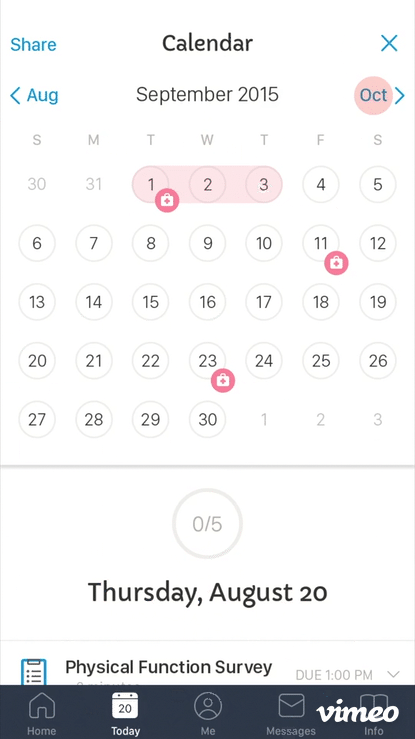
Scheduling and reminder systems were designed to integrate naturally into daily routines. Progress visualization gave patients a sense of forward momentum. Trial information — what the study was about, why it mattered, what their data was contributing — was woven throughout rather than buried in onboarding.
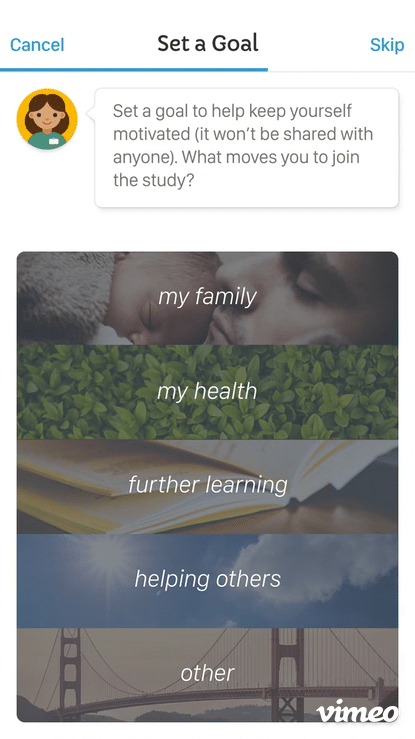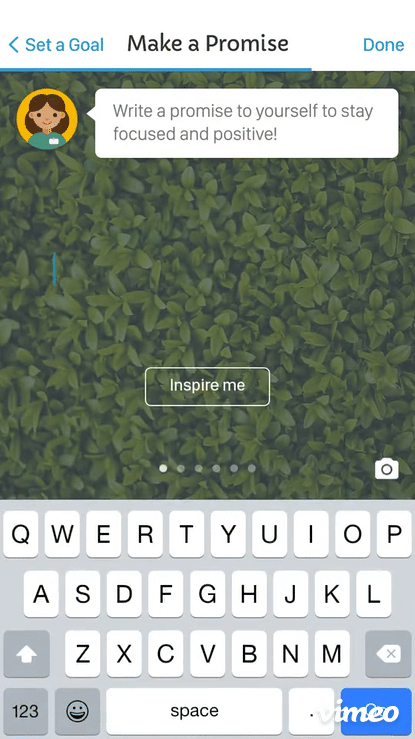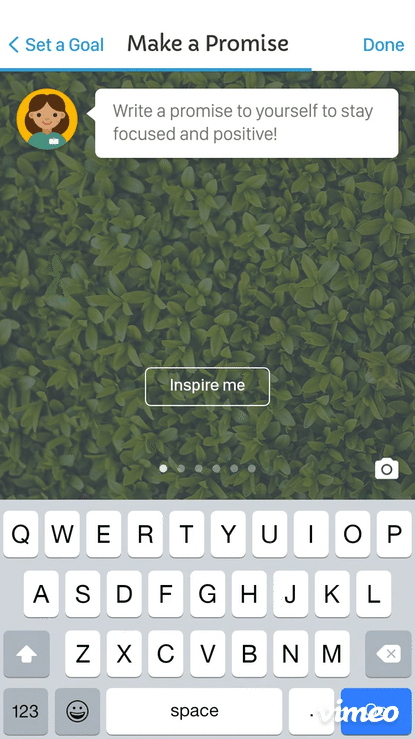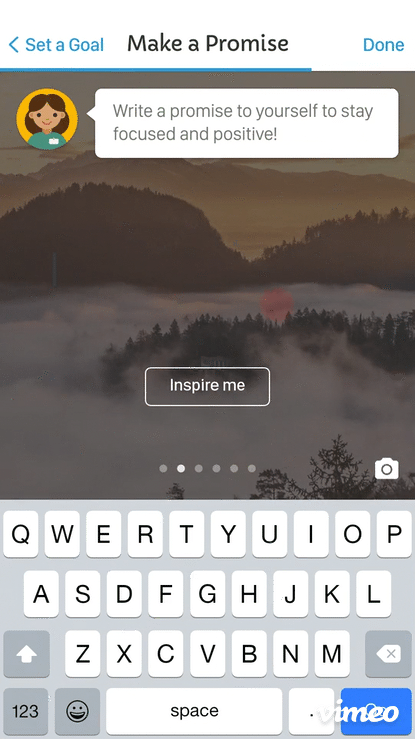
The motivational layer was the most nuanced design challenge: rather than generic encouragement, the experience was designed to reflect each patient's own stated reasons for participating — keeping their North Star visible across the full arc of the trial.
Outcome
The initiative was recognised with a Clinical Partnership of the Year award and a Scrip Award nomination — two of the most significant accolades in clinical research. The work contributed directly to Medidata's positioning as a leader in patient-centered clinical technology and informed the company's broader mobile health strategy across its global client base.
Most importantly, it demonstrated that when clinical trials are designed around the people inside them, participation doesn't have to be something patients endure — it can be something they commit to.
Full design documentation available on request.
Patient-Centric Strategy
My Role — UX Strategist & Lead Designer, Mobile Health Initiative · Medidata Solutions · 3 years · Deliverables: mobile health strategy, patient engagement platform, behavioral design framework, user research program, design system
Industry Recognition — Clinical Partnership of the Year · Scrip Award Nomination
The Problem
Clinical trial participation places enormous demands on patients — managing complex medication schedules, completing surveys, attending visits, and staying engaged across months or years — yet the tools built to support them had always been designed for institutions, not people. The result was persistent and measurable: poor adherence, high dropout, and a fundamental disconnect between what trials needed from patients and what patients needed to keep going.

The Research Approach
I designed a research programme that went beyond the industry convention of five-user testing, drawing on evidence that small samples miss significant proportions of real user problems. Research ran across three groups simultaneously: patients managing ongoing medication regimes, site personnel including nurses, physicians, and CRAs, and sponsors and medical directors overseeing large multi-site trials.

From the research I produced affinity maps, user journey maps, detailed archetypes, and a comprehensive app map with feature recommendations. Across all groups, a consistent pattern emerged: patients who sustained engagement had found a personal reason for being in the trial — a North Star — that the existing product did nothing to surface or reinforce.



The Design Framework
The research produced a behavioural design framework built around four interconnected pillars: Motivation, Scheduling, Activities, and Feedback.


The most important insight was about motivation specifically. Patients who sustained engagement weren't driven by abstract altruism — they had set a personal North Star at the outset. The design needed to surface and reinforce that goal throughout the experience, not just at onboarding. Five additional principles emerged from the research to guide every subsequent design decision:
— Activity as progress: patient effort needed to feel like achievement, not compliance — Forward-looking over backward-looking: where they were going mattered more than what they'd already done — Schedules afford habits: structure and routine were the primary mechanisms for sustained participation — Actionable guidance: bite-size tasks reduced cognitive burden and increased completion — Trial information builds trust: patients who understood what they were contributing stayed more engaged
The Design Response
These principles shaped a mobile experience designed to feel like a supportive companion rather than a monitoring tool. The app centred on a daily task view that gave patients a clear, achievable picture of what was needed from them that day — without overwhelming them with the full weight of the trial ahead.

Scheduling and reminder systems were designed to integrate naturally into daily routines. Progress visualization gave patients a sense of forward momentum. Trial information — what the study was about, why it mattered, what their data was contributing — was woven throughout rather than buried in onboarding.

The motivational layer was the most nuanced design challenge: rather than generic encouragement, the experience was designed to reflect each patient's own stated reasons for participating — keeping their North Star visible across the full arc of the trial.

Outcome
The initiative was recognised with a Clinical Partnership of the Year award and a Scrip Award nomination — two of the most significant accolades in clinical research. The work contributed directly to Medidata's positioning as a leader in patient-centered clinical technology and informed the company's broader mobile health strategy across its global client base.
Most importantly, it demonstrated that when clinical trials are designed around the people inside them, participation doesn't have to be something patients endure — it can be something they commit to.
Full design documentation available on request.
Patient-Centric Strategy
My Role — UX Strategist & Lead Designer, Mobile Health Initiative · Medidata Solutions · 3 years · Deliverables: mobile health strategy, patient engagement platform, behavioral design framework, user research program, design system
Industry Recognition — Clinical Partnership of the Year · Scrip Award Nomination
The Problem
Clinical trial participation places enormous demands on patients — managing complex medication schedules, completing surveys, attending visits, and staying engaged across months or years — yet the tools built to support them had always been designed for institutions, not people. The result was persistent and measurable: poor adherence, high dropout, and a fundamental disconnect between what trials needed from patients and what patients needed to keep going.

The Research Approach
I designed a research programme that went beyond the industry convention of five-user testing, drawing on evidence that small samples miss significant proportions of real user problems. Research ran across three groups simultaneously: patients managing ongoing medication regimes, site personnel including nurses, physicians, and CRAs, and sponsors and medical directors overseeing large multi-site trials.

From the research I produced affinity maps, user journey maps, detailed archetypes, and a comprehensive app map with feature recommendations. Across all groups, a consistent pattern emerged: patients who sustained engagement had found a personal reason for being in the trial — a North Star — that the existing product did nothing to surface or reinforce.



The Design Framework
The research produced a behavioural design framework built around four interconnected pillars: Motivation, Scheduling, Activities, and Feedback.


The most important insight was about motivation specifically. Patients who sustained engagement weren't driven by abstract altruism — they had set a personal North Star at the outset. The design needed to surface and reinforce that goal throughout the experience, not just at onboarding. Five additional principles emerged from the research to guide every subsequent design decision:
— Activity as progress: patient effort needed to feel like achievement, not compliance — Forward-looking over backward-looking: where they were going mattered more than what they'd already done — Schedules afford habits: structure and routine were the primary mechanisms for sustained participation — Actionable guidance: bite-size tasks reduced cognitive burden and increased completion — Trial information builds trust: patients who understood what they were contributing stayed more engaged
The Design Response
These principles shaped a mobile experience designed to feel like a supportive companion rather than a monitoring tool. The app centred on a daily task view that gave patients a clear, achievable picture of what was needed from them that day — without overwhelming them with the full weight of the trial ahead.

Scheduling and reminder systems were designed to integrate naturally into daily routines. Progress visualization gave patients a sense of forward momentum. Trial information — what the study was about, why it mattered, what their data was contributing — was woven throughout rather than buried in onboarding.

The motivational layer was the most nuanced design challenge: rather than generic encouragement, the experience was designed to reflect each patient's own stated reasons for participating — keeping their North Star visible across the full arc of the trial.

Outcome
The initiative was recognised with a Clinical Partnership of the Year award and a Scrip Award nomination — two of the most significant accolades in clinical research. The work contributed directly to Medidata's positioning as a leader in patient-centered clinical technology and informed the company's broader mobile health strategy across its global client base.
Most importantly, it demonstrated that when clinical trials are designed around the people inside them, participation doesn't have to be something patients endure — it can be something they commit to.
Full design documentation available on request.